$1m fine for misleading Nurofen claims
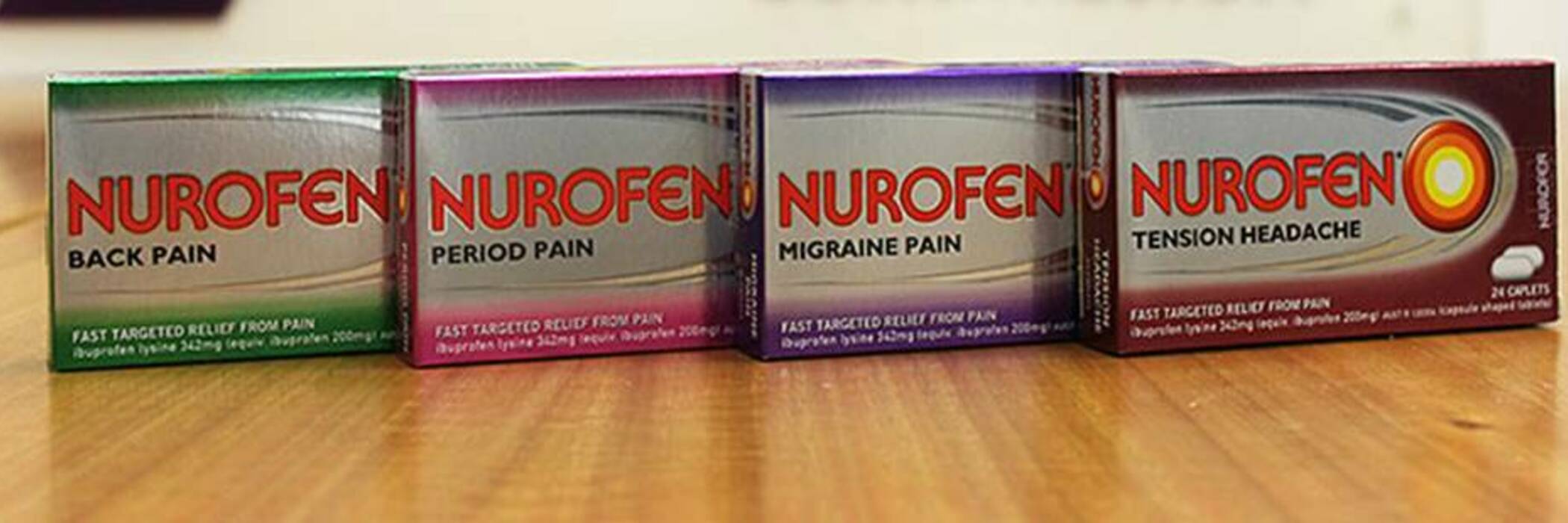
Each product claimed to target a particular type of pain.
Nurofen manufacturer Reckitt Benckiser (New Zealand) has copped a $1.08 million fine for 10 charges relating to its Nurofen specific pain range products.
The company admitted that between 2011 and 2015 product packaging and website representations were liable to mislead consumers about the nature, characteristics and suitability of the products.
Packaging claimed the products targeted a particular type of pain. However, each product contained exactly the same active ingredient, 342 mg ibuprofen lysine, and worked identically.
The products included Nurofen Migraine Pain, Nurofen Tension Headache, Nurofen Period Pain and Nurofen Back Pain.
“Consumers paid significantly more for these products compared to other ibuprofen products that would have had a similar effect,” Commerce Commission Chairman Dr Mark Berry said.
Dr Berry said the company was made aware in 2011 and 2013 through a media article and a complaint to the Australian Therapeutic Goods Administration that the products were potentially misleading but continued to market and sell them.
“We take a particularly dim view when goods for human consumption are misdescribed; especially where pharmaceutical or healthcare products are not promoted truthfully. With these types of products consumers have little opportunity to verify the claims being made and tend to rely heavily on what they are told by the trader. To be able to choose the product best suited for them, consumers must have accurate and reliable information,” said Dr Berry.
Judge Jelas described Reckitt Benckiser’s behaviour as “highly misleading” and noted it was “blatantly apparent they were in breach of their lawful obligations to New Zealand consumers”.
The judge accepted the company had expressed remorse for its conduct but only after the commission opened its investigation, having ignored previous warnings and publicity in the media.
Member comments
Get access to comment